News
Blog
About the future of product creation
Read more about
Model-Based Definition: Take Your Engineering Processes to the 21st Century
Read more about
Automated Offline Programming: The Future of Robotic Manufacturing
Read more about
Implementing Simulation-Driven Design: Five Tips for a successful Start
Read more about
PLM system integration for greater market success in mechanical engineering
Read more about
How can you achieve end-to-end SAP and 3DEXPERIENCE processes?
Read more about
Offline programming with the aid of a simulation tool
Read more about
Unleashing Business Success with Smart Application Management
Read more about
Leveraging data and transparency to reduce cost risks in engineering change management
Read more about
Six trends in Digital Product Development
Read more about
Design, modeling and simulation: A unified entity that needs differences
Read more about
How to leverage sustainability to make your business more innovative
Read more about
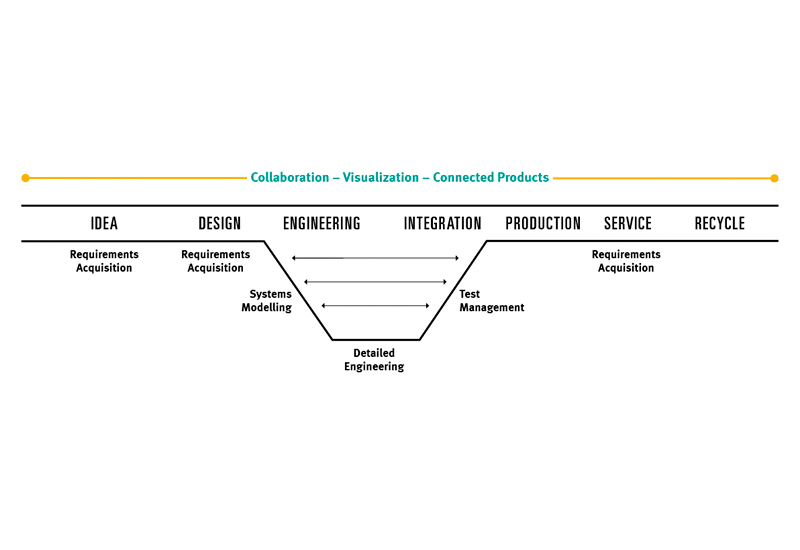
Transforming Product Creation
Read more about
How to optimize your operational excellence by working on ASPICE
Read more about
Data continuity in manufacturing industry: How software and IT are changing
Read more about
Digital Collaboration
Read more about
How will digitalization change collaboration in your manufacturing company?
Read more about
Digital Twins: Robot optimization in a matter of hours
Read more about
Hybrid Manufacturing
Read more about
You want to introduce PLM? Limited internal resources should not be an obstacle!
Read more about
Digital Production: Data Continuity from Design to Manufacturing
Read more about
Meissner relies more on robots
Read more about
CAD data management
Read more about
Geometric Product Specifications: Standards enable digital continuity and raise efficiency
Read more about
Leveraging SAP EPD Engineering to integrate requirements management and system modeling into your PLM process
Read more about
SaarGummi Automotive: A Smart IT Concept for Global Product Design
Read more about
Listening Improves Artificial Intelligence!
Read more about
What digital platform does my business need?
Read more about
Home Office for Robot Operators?
Read more about
Tolerance Analysis: From Excel to Digital Quality Twin
Read more about
The Digital Twin: It’s all about the Context
Read more about
Tolerance management: Digital twin reduces product lifecycle costs